I’m just back from Asia, where I had some good discussions with customers. One of questions I am commonly asked involves the long-standing issue of, “What do we mean by clean?” A lot of research has been done to try to define “clean,” usually using some measure of the level of contaminant: dirt, oxide, or flux residue, which is commonly referred to as “contaminating material” or CM. Test methods such as ROSE, surface extract resistivity, SIR, reflectance IR, are often employed, but in many instances, these tests lack either sufficient sensitivity or relevance to the actual failure mode caused by the CM.
The problem with these definitions using an analytical methodology is that they focus too much on quantifying the level of CM remaining after a cleaning process, leaving the second question unanswered until another day: “How low a level of CM is low enough?” This seems to be putting the cart before the horse.
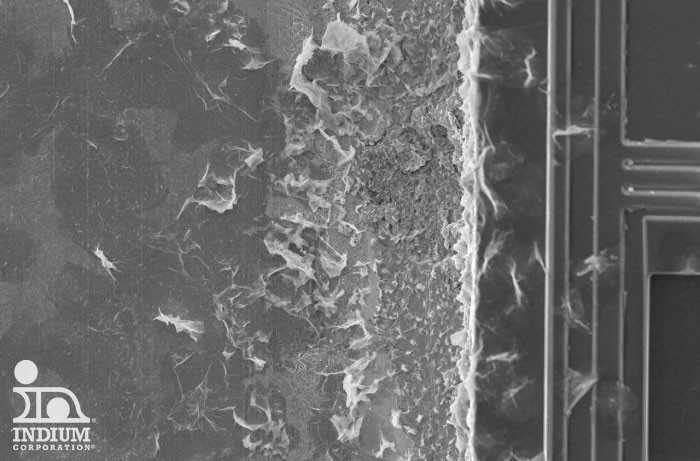
SEM is an excellent example of a cleanliness-determining test method that can be used to overanalyze for CM. Shown here is a picture of a perfectly acceptable no-clean solder paste residue shown at very high magnification under SEM. Note that it is difficult to avoid the tendency to want to ask "what is this thing here? And what about this thing?" It's more important to know that billions of successful devices have been built with no failures due to CM, and that also conform to the most rigorous automotive reliability test standards.
Rather than focusing on the CM, we should be looking at the failure mode caused by the residue. In this way we can give a definition of clean that will be relevant to the situation. For example, CM may cause failures such as wirebonding pull strength, electrochemical migration (ECM) or delamination. In each case, the failure can be quantified. There’s a well proven principle that “a difference that makes no difference is not a difference,” and we can apply that rigorously to our attempt to define “clean.”
We begin by determining a clear metric for the extent or rate of occurrence (ppm or %) of the failure caused by the CM. Once that is done, we can establish a clear and unambiguous definition: “If there is no statistically significant difference in the failures seen between (1) an uncontaminated sample, and (2) a previously contaminated sample that has been subsequently cleaned, then the cleaning process is good enough.” This will depend very much on your chosen % confidence level, but it provides a pragmatic way of defining cleanliness easily and unambiguously, and effort can then be spent on establishing a “good enough” cleaning protocol, rather than an arbitrary chase to zero contaminant.
Process monitoring of a key control variable can then be established that correlates with the failure mode. This could be things such as: cleaning bath contamination (possibly using the cleaning chemical concentration, turbidity, or electrical resistance), contact angle, surface reflectance IR, SEM/EDX, and so on. Usually establishing this key variable and a sampling rate, and also ensuring the sample and test method are representative, are not easy, and it is probably this complexity that leads to the tendency to focus on this latter task as the more important of the two.
Cheers! Andy