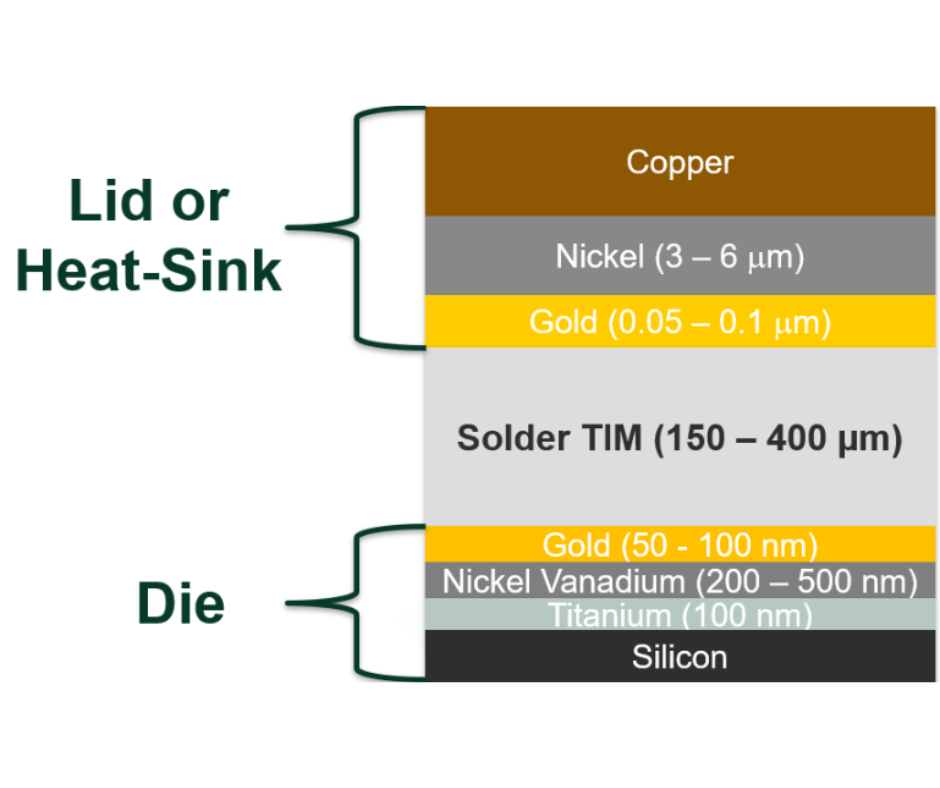
Generically, a phase change material is one which will store or release energy when it changes phase from solid to liquid or liquid to solid.According to this generic classification, there are 4 general categories of phase change materials.
- Salt Hydrates such as Sodium Sulfate, Calcium Chloride, Sodium Acetates,etc
- Eutectic Salts
- Paraffins
- Non-Paraffin Organics
These phase change material categories are not all-encompassing, however. Other materials such as metals, eutectic or not, are used as phase change materials for their thermal energy storage and removal abilities.
Nearly all soft solders classify as phase change materials according to their melting temperature.According to Maurice J. Marongiu from MJM Engineering, who conducted a webinar on phase change materials, the melting temperature of a typical phase change material is between 0-250ºC.Solders officially may melt at higher temperatures, such as the AuGe eutectic alloy which melts at 356ºC, however the majority of solders used melt below 250ºC.
Phase change materials are a common occurrence in the world of thermal interfaces for electronics.Here, tighter commonalities between phase change materials can be found.For instance, the phase change temperature for these thermal interface materials is within the range of a common TIM junction temperature, which is typically lower than 100ºC.For this reason, when we consider a metal interface to be a phase change material in this industry, it is an alloy or material which changes phase below 100ºC.
When implementing a metal or non-metal phase change material into a thermal interface, there are some design considerations to be made:
- Phase change materials are applied as solid pads.At room temperature they are firm and available with specific dimensions which make them easy to handle.Consistent application should be inherent.
- Phase change materials each change phase at a unique temperature.The appropriate phase change material engineered for an application will have a phase change temperature reached within the normal operating cycle of the device.
- Phase change materials are designed to turn liquid in operation.The liquid phase of these materials will have a distinct viscosity.Depending on the material, clamping pressure and assembly orientation, the molten material may leak.Proper precautions should be taken to prevent material leakage, especially toward active electrical components if the phase change material is electrically conductive.
- When reservoirs are created to contain a phase change material, these reservoirs must accommodate the liquid phase of the phase change material as well as the solid phase.As a phase change material changes from solid to liquid there is an increase in the material volume.If the phase change material expands and fractures the reservoir, this will lead to leaks and the eventual failure of the electronic device as the thermal interface becomes backfilled with air.