At times, I get questions like, “How long does it take to deposit 20 µm of Indium on my plate using your Indium Sulfamate Bath?” Well, the answer to this question would make it very easy for the customer. Let me show you how to calculate this with ease. In this post, I will derive a simple and easy relation that will work for anyone. Believe me, it’s easy; just use some fundamental relations that we studied in high school. It starts with how many electrons you require to reduce an In3+ ion to an In atom – 3, right!
Quindi, quanto è la carica - 3e!
The time it takes directly depends upon how much charge we’ve got to supply to reduce all those Indium ions, and at what time rate we can supply those – that is “current”!
If you’ve heard of this famous current-charge relation, i =q/t, yes, that is what I am talking about.
Quindi, torniamo alle basi,

If the cathode (where the indium is electrodeposited) is not 100%efficient, it will take a little longer. Let’s say, σ be the efficiency. Then it will take, 1/σtimes longer. For example, if it is50% efficient, it will take, 1/0.5 = 2times longer. So, incorporating efficiency, the equation for time becomes:

Inserendo tutti questi valori nell'equazione precedente, si ottiene:

Okay, so the answer is, the estimated time to deposit 20µm is 34 minutes, and you know what factors can change this.
And here’s an easy formula to calculate time in minutes for the most widely used units of thickness and current density:
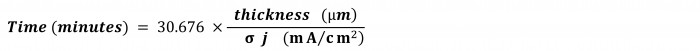
Ok, perché non chiamare questo numero 30.676 "costante di Inplate di Shital"!
Indium Sulfamate Bath from Indium Corporation has a cathodic efficiency of 90%. More efficiency means less time! The typical current density to work on is around 10-20mA/cm2but it can be increased up to100 mA/cm2, maintaining the bath temperature at 20-25°C. Increasing the current density from 20mA/cm2to100 mA/cm2can reduce the deposition time by a factor of 5, i.e. the same 20µm will now deposit in 7 minutes.
The extensive use of indium plating today dates back to the 1930s when the founders of Indium Corporation developed a commercial indium plating bath for the first time. Recent developments in semiconductor technology and flip-chip bonding utilize indium to create interconnects between layers of wafers. Indium is electroplated onto wafer substrates to create high-density, low-pitch and high-aspect ratio indium bumps. The softness, ductility and wettability of indium ensure a strong and reliable connection between two surfaces, even if they are not perfectly flat or aligned. Furthermore, Indium exhibits stability even at temperatures approaching zero, making it highly suitable for use in semiconductor applications that operate in extreme environments, such as those encountered in space.
Se state pensando a progetti di galvanotecnica su piccola scala, tra cui il restauro e la riparazione di oggetti metallici antichi con rivestimento all'indio, potete iniziare con un kit di galvanotecnica sano e facile da usare come quello offerto qui.
Per saperne di più sulla galvanizzazione dell'indio, sul bagno di solfammato di indio e sulla galvanizzazione di bump di indio, consultare i seguenti documenti.
- Ottenere una struttura dei grani più fine utilizzando il bagno di placcatura al solfammato di indio
- Indium Bump Electroplating
- Placcatura, un metodo alternativo di applicazione dell'indio
- Preparazione corretta della superficie per la placcatura con indio
- Placcatura di prototipi con bagno di placcatura al solfammato di indio
- Bonifica e smaltimento della soluzione del bagno di placcatura al solfammato di indio
For any other inquiries, please contact me.



