At times, I get questions like, “How long does it take to deposit 20 µm of Indium on my plate using your Indium Sulfamate Bath?” Well, the answer to this question would make it very easy for the customer. Let me show you how to calculate this with ease. In this post, I will derive a simple and easy relation that will work for anyone. Believe me, it’s easy; just use some fundamental relations that we studied in high school. It starts with how many electrons you require to reduce an In3+ ion to an In atom – 3, right!
그렇다면 요금은 얼마인가요 - 3e!
The time it takes directly depends upon how much charge we’ve got to supply to reduce all those Indium ions, and at what time rate we can supply those – that is “current”!
If you’ve heard of this famous current-charge relation, i =q/t, yes, that is what I am talking about.
이제 기본으로 돌아가 보겠습니다,

If the cathode (where the indium is electrodeposited) is not 100%efficient, it will take a little longer. Let’s say, σ be the efficiency. Then it will take, 1/σtimes longer. For example, if it is50% efficient, it will take, 1/0.5 = 2times longer. So, incorporating efficiency, the equation for time becomes:

위의 방정식에 이 모든 값을 입력하면 다음과 같은 결과를 얻을 수 있습니다:

Okay, so the answer is, the estimated time to deposit 20µm is 34 minutes, and you know what factors can change this.
And here’s an easy formula to calculate time in minutes for the most widely used units of thickness and current density:
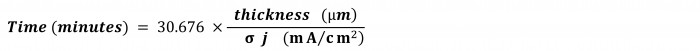
좋아요, 이 숫자를 30.676 "시탈의 인플레이트 상수"라고 부르면 어떨까요?
Indium Sulfamate Bath from Indium Corporation has a cathodic efficiency of 90%. More efficiency means less time! The typical current density to work on is around 10-20mA/cm2but it can be increased up to100 mA/cm2, maintaining the bath temperature at 20-25°C. Increasing the current density from 20mA/cm2to100 mA/cm2can reduce the deposition time by a factor of 5, i.e. the same 20µm will now deposit in 7 minutes.
The extensive use of indium plating today dates back to the 1930s when the founders of Indium Corporation developed a commercial indium plating bath for the first time. Recent developments in semiconductor technology and flip-chip bonding utilize indium to create interconnects between layers of wafers. Indium is electroplated onto wafer substrates to create high-density, low-pitch and high-aspect ratio indium bumps. The softness, ductility and wettability of indium ensure a strong and reliable connection between two surfaces, even if they are not perfectly flat or aligned. Furthermore, Indium exhibits stability even at temperatures approaching zero, making it highly suitable for use in semiconductor applications that operate in extreme environments, such as those encountered in space.
인듐 코팅이 된 골동품 금속 제품의 복원 및 수리를 포함한 소규모 전기 도금 프로젝트를 생각하고 있다면 여기에서 제공하는 것과 같이 사용하기 쉬운 전기 도금 키트로 시작할 수 있습니다.
인듐 전기 도금, 인듐 설파메이트 배스 및 인듐 범프 전기 도금에 대해 자세히 알아보려면 다음 문서를 참조하세요.
- 황산인듐 도금조를 사용하여 더 미세한 입자 구조 달성하기
- Indium Bump Electroplating
- 도금, 인듐 도포의 대체 방법
- 인듐 도금을 위한 적절한 표면 준비
- 황산인듐 도금조를 사용한 프로토타입 도금
- 황산인듐 도금 수조 용액의 회수 및 폐기
For any other inquiries, please contact me.



